Battery capacity quantifies how much charge a battery can store and deliver—making it a critical metric for applications ranging from smartphones to electric vehicles. While the intuitive definition of capacity may seem straightforward, there are multiple ways to measure and report it.Documentation Index
Fetch the complete documentation index at: https://docs.ionworks.com/llms.txt
Use this file to discover all available pages before exploring further.
Types of Capacity
Nominal Capacity
The most common definition of capacity is nominal capacity, the value typically provided by the battery’s manufacturer. This is the amount of charge (measured in ampere-hours, Ah) the battery is designed to deliver under standardized test conditions.Nominal capacity serves as a benchmark for comparing different batteries, but
it represents an idealized value—hence it’s often reported as a round number.
Real-world conditions (temperature fluctuations, aging, faster discharge
rates) usually result in lower usable capacity.
C-Rate Specific Capacity
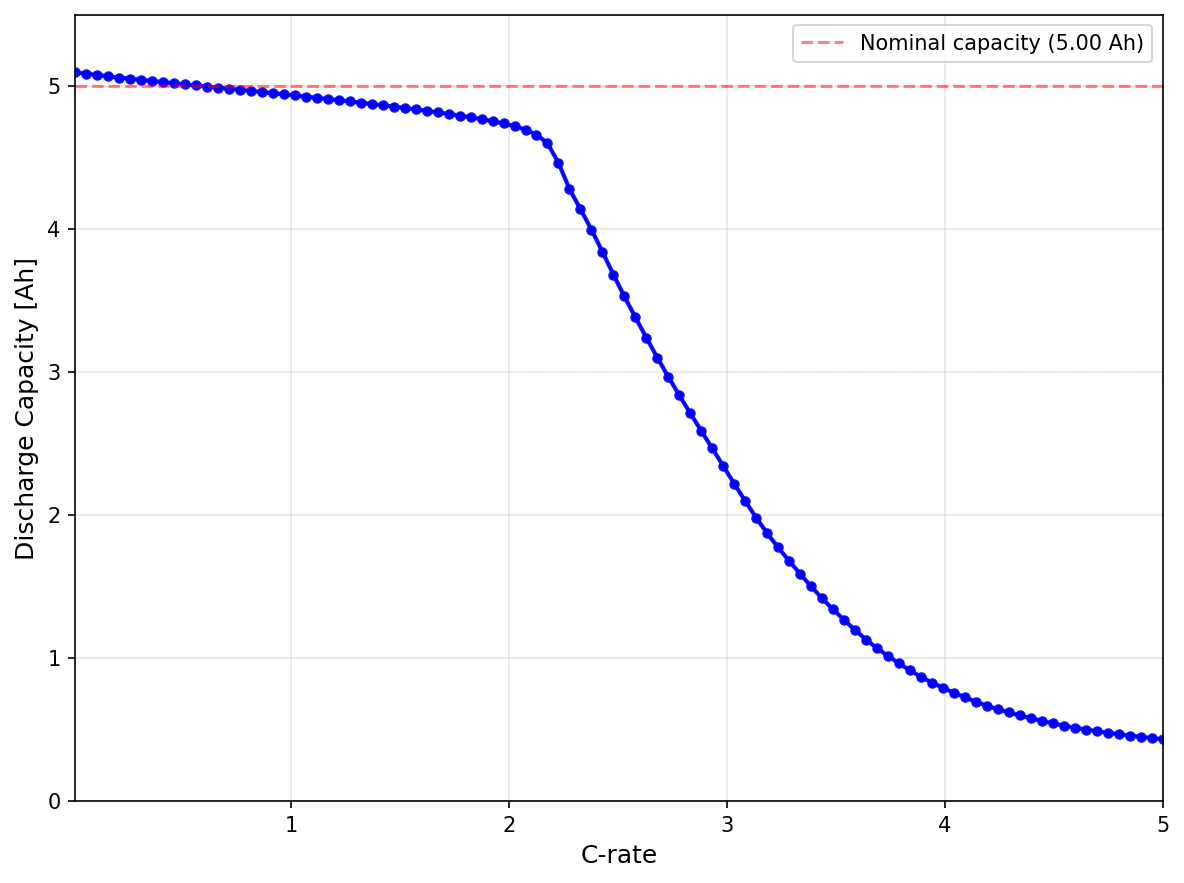
The C-rate defines how quickly a battery is charged or discharged relative to its nominal capacity. A C-rate of 1C means the battery would be fully discharged in 1 hour; 2C means 30 minutes; C/2 (or 0.5C) means 2 hours. The current corresponding to a given C-rate is: For example, a 50 Ah battery at 2C draws A.| C-Rate | Discharge Time | Typical Application |
|---|---|---|
| C/20 | 20 hours | Capacity testing, characterization |
| C/3 | 3 hours | Standard capacity measurement |
| 1C | 1 hour | Typical EV discharge |
| 2C | 30 minutes | Fast discharge, high power |
Measuring Capacity at a Specific C-Rate
To measure capacity at a given C-rate:- Fully charge the battery using the manufacturer’s recommended protocol
- Rest until voltage stabilizes (typically 1-4 hours)
- Discharge at constant current corresponding to the desired C-rate
- Stop when the lower voltage cutoff is reached
- The capacity equals the integrated current over time
Theoretical Capacity
If we could discharge a battery infinitely slowly, we would obtain the theoretical (or thermodynamic) capacity. This is the maximum charge a battery can deliver within a given voltage window (e.g., 4.2 V to 2.5 V) when discharged infinitely slowly, eliminating all kinetic and transport limitations. In practice, we cannot discharge a battery infinitely slowly, so we approximate the theoretical capacity by discharging at a very low C-rate. This will always result in an underestimation of the true theoretical capacity.Related Topics
- State of Charge—measuring remaining charge relative to capacity
- State of Health—how capacity fades over time
- Internal Resistance—why capacity decreases at high C-rates
- Reaction Kinetics—the physics behind rate-dependent capacity loss
- Degradation Overview—mechanisms that cause capacity fade